Quick Facts
Nickel Alloy 400 is a nickel copper alloy which has high strength over a wide temperature range up to 1000 F. It is regarded as being a ductile Nickel-Copper alloy with resistance to a wide variety of corrosive conditions. This alloy is most frequently applied in a range of environments going from mildly oxidizing through neutral and to moderately reducing conditions. Additional application area of this material is in marine environments and other non-oxidizing chloride solutions. Like with commercially pure Nickel, Monel® 400 is low in strength in the annealed condition, for this reason a variety of tempers are used to achieve higher strength levels. Nickel Alloy 400 is one of the few alloys that maintains its strength in sub-zero or cryogenic temperatures, so it is often used in those applications.
Typical Applications
- Valves
- Pumps
- Fasteners
- Fittings
- Heat exchangers
Stock Range
We stock a range of round bars, sizes between 12.7 mm and 50.80mm in diameter, no Flat Bars
Primarily manufactured in Europe or US,
Melting Practices
- Induction furnace
Chemical Analysis

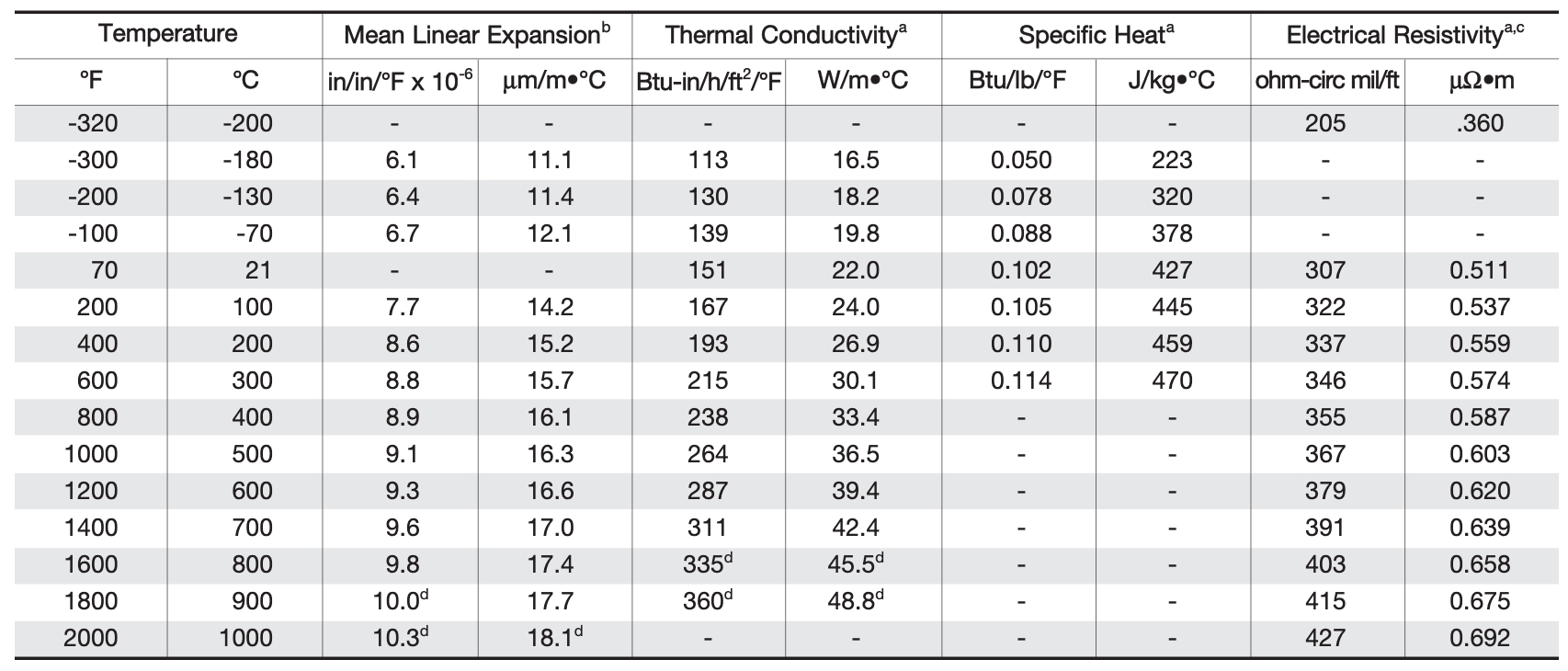
Mechanical Properties
Machinability
Alloy 400 can be machined at satisfactory rates with machine tools generally employed by industry. In general, cold-drawn or cold-drawn, stress-relieved materials recommended for best machinability and smoothest finish. Nickel alloy R-405 is the free-machining version of Nickel alloy 400.
Corrosion Resistance
Nickel Alloy 400 exhibits resistance to corrosion by many reducing media. It is also generally more resistant to attack by oxidizing media than higher copper alloys. This versatility makes alloy 400 suitable for service in varies environments. Alloy 400 is widely used in marine applications. While alloy 400 products exhibit very low corrosion rates in seawater, stagnant conditions have been shown to induce crevice and pitting corrosion. Alloy 400 is also resistant to stress corrosion cracking and pitting in most fresh and industrial waters.
MONEL alloy 400 offers exceptional resistance to hydrofluoric acid in all concentrations up to the boiling point. It is perhaps the most resistant of all commonly used engineering alloys. Alloy 400 is also resistant to many forms of sulfuric and hydrochloric acids under reducing conditions.

